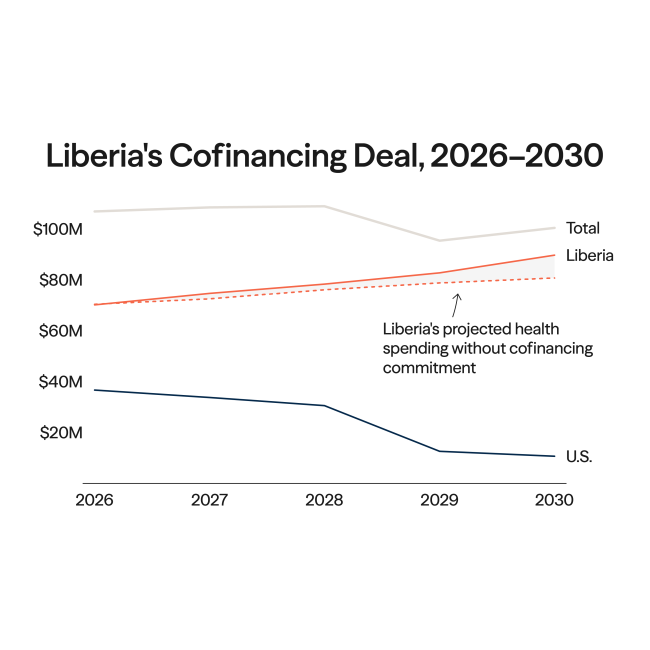
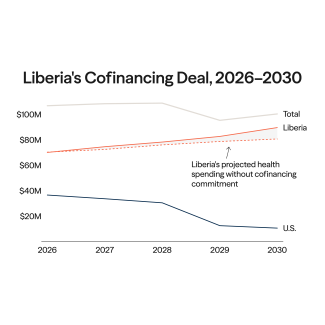
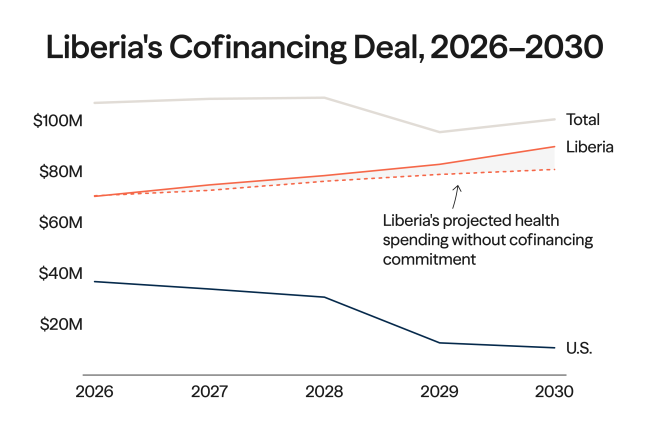
Indonesia joined the BRICS, an intergovernmental organization composed of emerging economies, on January 7, 2025, at the start of a year that saw global health governance thrown into flux. The abrupt dissolution of the U.S. Agency for International Development (USAID), health overseas development assistance (ODA) reaching a 10-year low, and adoption of the World Health Organization's (WHO's) Pandemic Agreement created an opening to reshape multilateralism and shift from established paradigms centered around foreign aid to more equitable participation between high- and low- or middle-income countries (LMICs). Indonesia's accession to the BRICS, therefore, should be read as a pragmatic attempt to widen its negotiating avenue and reduce its dependency, particularly in pharmaceuticals research and development (R&D) and health emergency preparedness.
Indonesia's accession to the BRICS should be read as a pragmatic attempt to widen its negotiating avenue and reduce its dependency
BRICS members offer fresh perspectives to the established market-driven, private-sector-oriented model. The bloc's emphasis on strengthening human capital, through expanding access to affordable medicines and building domestic pharmaceutical capacity, complements Indonesia President Prabowo Subianto's National Transformation Strategy to improve infrastructure and strengthen the country's economy. By joining BRICS, Indonesia can take advantage of existing diplomatic levers, infrastructure, and manufacturing to unlock a pathway to locally producing end-to-end priority drugs and establishing a major role in global health. These shared priorities align with BRICS initiatives on tuberculosis (TB) vaccines and treatment R&D, offering Indonesia domestic gains and an opportunity to expand South-South health cooperation.
Global Health Cooperation from BRICS
To determine the extent to which Indonesia's decision to join the BRICS can influence global health, it is important to look at other members' engagement in health. Since joining the bloc in 2009, Brazil has played a leading role in global health-policy negotiations, including providing antiretroviral drugs for people living with HIV/AIDS and expanding access to TB drugs. In 2025, Brazil's BRICS presidency spotlighted a critical juncture to end TB by 2030. The country convened the BRICS Tuberculosis Research Network, which aims to develop new vaccines and accelerate production of treatments and vaccines against the disease. Brazilian institutions Fiocruz and Bio-Manguinhos developed the first national messenger RNA (mRNA) platform for vaccines and therapies to be used against TB. That network is particularly valuable to Indonesia, which has the second largest TB incidence worldwide.
China, through its Belt and Road Initiative, has engaged in vaccine diplomacy, exemplified by the rapid and extensive distribution of Chinese-produced COVID-19 vaccines. Since 2021, Indonesia's bilateral agreements with China have allowed for collaboration in the vaccine and genomics ecosystem, and in 2026, the two countries committed to establishing artificial intelligence (AI) in medicine. Although Indonesia maintains bilateral health ties with China, BRICS membership offers the country access to the Vaccine R&D Center, which was presented by China in 2022. The center now supports Indonesia's Group of 20 (G20) Presidency agenda on equitable research and production of vaccines.

India, a BRICS member that supplies 20% of all generic drugs prescribed globally, has long been known as the "pharmacy of the world." For Indonesia, greater proximity to India's pharmaceutical ecosystem could translate into opportunities to deepen existing collaboration. Most immediately, Indian manufacturers are positioned to transform chronic disease treatment through biosimilar semaglutide, a GLP-1 receptor agonist used to treat type 2 diabetes and obesity. Indian manufacturers are expected to produce semaglutide for less than $6 per month following the May 2026 patent expiration in Brazil, Canada, China, and India—far less than branded products like Wegovy, which costs $195 (3.1 million Indonesian rupiah) per month in Indonesia.
Indonesia's Path
Indonesia can negotiate advance-procurement agreements to import biosimilar semaglutide once production scales, improving access for millions of Indonesians currently priced out of treatment. This price difference matters for Indonesia, where diabetes prevalence among individuals aged 15 years and older increased from 10.9% to 11.7% between 2018 and 2023 and among adults living with overweight or obesity from 35.4% to 37.8% in the same period.
Collaboration in chronic disease treatment could build on the already operational India-Indonesia pharmaceutical partnership. Since 2011, Bio Farma has collaborated with the Serum Institute of India on vaccine development and access. In January 2025, the two companies formalized a partnership [PDF] on tuberculosis diagnostics and recombinant BCG vaccine development. BRICS membership strengthens this collaboration by situating it within wider manufacturing and R&D initiatives instead of just part of a standalone bilateral-collaboration track, and strengthens Indonesia's position to leverage India's manufacturing scale for future vaccine development.
In December 2025, Minister of Industry Agus Gumiwang Kartasasmita reaffirmed Indonesia's commitment to collaborate with Russia through the BRICS Centre for Industrial Competencies (BCIC), which encompasses bioindustry development alongside other key areas. Within bioindustry, oncology stands out as particularly advantageous: Russia's Gamaleya National Research Center, in collaboration with the National Medical Research Radiological Centre, has developed a personalized mRNA-based cancer vaccine (Neoonkovac) that received approval for clinical use on November 21, 2025.
Although the vaccine is approved for melanoma, the adaptability of mRNA platform technology for other cancers in the future could be strategic for Indonesia. In 2022, the Global Cancer Observatory [PDF] recorded more than 400,000 new cancer cases in Indonesia, with a mortality rate exceeding 50%. The next year, the Ministry of Health Indonesia published the National Cancer Strategy 2024-2034 [PDF], including the cost analysis: cancer is the second most expensive catastrophic disease in Indonesia and accounts for 17% of Indonesia's national health-insurance catastrophic diseases budget, costing approximately 5.97 trillion Indonesian rupiah ($35.86 million) in 2023 alone.
BRICS membership could give Indonesia preferential access to licensing or coproduction arrangements for Russia's mRNA cancer vaccine technology, extending BCIC's bioindustry mandate into oncology and signaling Indonesia's intent to move up the pharmaceutical value chain beyond the manufacturing of generic products that it acquires from India.
Alternatives from the West
Beyond diplomacy, the BRICS also seeks influence in financing and aid models. The creation of the BRICS' New Development Bank (NDB) signaled a shift from traditional finance institutions like the World Bank and the seeking of new strategies to grow the bloc's influence and create a new order. For Indonesia, the NDB's financing is particularly attractive: 88.2% of loans [PDF] are government-to-government lending, 34.5% are denominated in local currency, and the bank operates without policy conditionalities [PDF] attached. This means that the government can access NDB financing to build domestic vaccine-manufacturing plants, pharmaceutical production facilities, and health R&D infrastructure without the reforms or restrictions typically attached to World Bank lending.
Indonesia can use its membership to bring fresh mediating skills to the BRICS negotiating table. As part of the Group for Equity, which is coleading discussions around the Pathogen Access and Benefit-Sharing (PABS) system annex to the WHO Pandemic Agreement, Indonesia could help unify BRICS countries and advance a more pragmatic, cohesive pandemic-preparedness strategy. Successfully brokering BRICS cohesion on PABS could elevate Indonesia's profile in global health governance, establishing the country as a credible negotiator in international health law. BRICS membership, combined with Indonesia's G20 experience, strengthens this positioning. On the other hand, if Indonesia limits its role to avoid straining relations with Western allies, it will undermine the self-reliant pharmaceutical strategy and financing autonomy that President Prabowo's National Transformation Strategy requires.
In the coming years, Indonesia will have to determine its participation in the bloc's health-cooperation dynamics. BRICS membership provides Indonesia with the partnerships, financing, and technology transfer pathways needed to build end-to-end pharmaceutical manufacturing capacity. Furthermore, BRICS's centers, such as the Vaccine R&D Center or the Center for Industrial Competencies, can complement bilateral cooperation by enabling scale, coordination, and bargaining leverage.