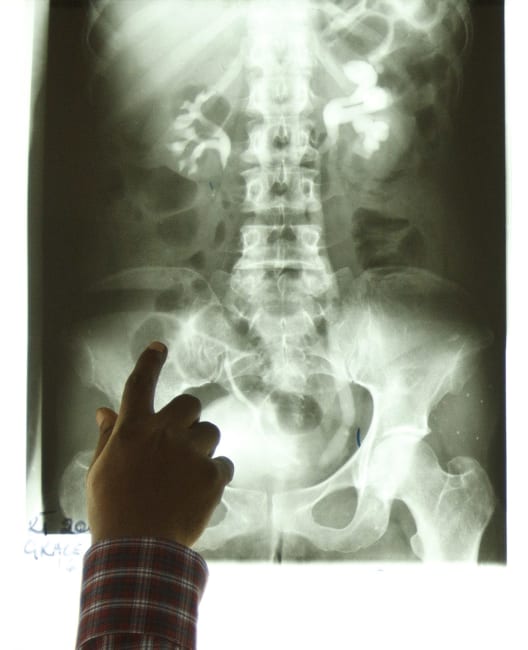
In 2020, almost 10 million people worldwide became sick with tuberculosis (TB) and 1.5 million died, making TB the second deadliest infectious disease behind COVID-19. Like COVID, TB is primarily a respiratory virus, but it can affect other parts of the body, including the kidneys and brain. It's caused by a bacterium called mycobacterium tuberculosis that spreads through the air from one person to another.
While most cases show no symptoms—called latent TB—about five to 10 percent of infected people develop active symptoms including a chronic cough marked by blood-containing mucus, chest pain, fever, and chills. People with HIV and diabetes—more fragile immune systems—are more susceptible to TB infection.
Tuberculosis disproportionately affects people in low- and middle-income countries, with 98 percent of reported cases in LMICs. For the first time in a decade, deaths from tuberculosis are on the rise. The scope and intensity of the global tuberculosis epidemic is in large part fueled by inadequate and antiquated TB drugs, most of which were developed more than 50 years ago.
Treating Tuberculosis
Treatment for active tuberculosis usually consists of a cocktail of antibacterial medications taken over the course of six to twelve months. Many TB patients typically show signs of improvement in the first few weeks after starting treatment, so they may stop taking their medications early, but this can lead to drug-resistant TB, multidrug resistant and rifampicin-resistant TB (MDR/RR-TB), or extensively drug-resistant tuberculosis (XDR-TB)—what type it is depends on which drug or drugs a case is resistant to. Poor prescribing practices and difficulty accessing medications can lead to forms of drug-resistant TB, as well.








Treating drug-resistant TB is particularly challenging and can take 20 to 30 months to complete. And patients often experience drug side effects, including nausea and vision issues. Even then, these complex and expensive treatments aren't very effective in the long term.
But researchers continue to seek new treatments that are shorter, more effective, and safer for patients. Recently, results from a TB Alliance clinical trial that treated 181 patients with highly-drug resistant TB in South Africa, Russia, Georgia, and Moldova, with two new drugs—bedaquiline (B) and pretomanid (Pa)—along with varying doses and durations of a third drug, linezolid (L), as part of a new treatment known as the BPaL, were encouraging and reported limited adverse side effects compared with traditional TB treatments. This month, another TB drug trial announced it has recruited patients from four continents.
EndTB, a partnership between Partners In Health (PIH), Doctors Without Borders, Interactive Research and Development, and UNITAID said that 750 patient volunteers from seven countries—Georgia, India, Kazakhstan, Lesotho, Pakistan, Peru, and South Africa—have enrolled in a phase 3 randomized controlled trial comparing five new treatment regimens for MDR-TB. They'll include two of three new TB drugs—bedaquiline and delamanid—in combination with existing oral TB drugs.